The reliance on borehole water necessitates a thorough understanding of its quality. Due to water shortage, many households in the city of Kara use water from boreholes directly supplied untreated in the dry and wet seasons. Therefore, this work aimed to investigate the chlorine demand of raw water from private boreholes and perform a spatiotemporal evaluation of the water quality in the city of Kara, Togo. Two sampling campaigns spread over dry (DS) and rainy (RS) seasons in April and August 2023, were carried out to capture seasonal and spatial differences across various boreholes (n=32) in the study area. The physicochemical parameters and germ indicators of fecal contamination were assessed via the standardized method (AFNOR) and WHO Guidelines for Drinking Water Quality (GDWQ). The correlation matrix analysis revealed a complex effect from the dissolution of local rocks (amphibolites, pyroxenites, etc.) and anthropogenic activities. Water was noncompliant concerning faecal contamination in 84.3% of the cases in the dry season (DS) and 96.37% of the cases in the rainy season (RS). The color, TH, TAC, turbidity, FeT, nitrate ion, and electrical conductivity values for some boreholes were outside the GDWQ values. Boreholes F8, F9, and F25 have very high Larson corrosion index (LR>1.2). The chlorine demand varies according to the season, ranging from 0.25 to 6.0 mg/L in DS and from 0.25 to 2.45 mg/L in RS. Regular monitoring is needed to ensure safe drinking water from boreholes in Kara.
| Published in | American Journal of Applied Chemistry (Volume 12, Issue 6) |
| DOI | 10.11648/j.ajac.20241206.14 |
| Page(s) | 158-172 |
| Creative Commons |
This is an Open Access article, distributed under the terms of the Creative Commons Attribution 4.0 International License (http://creativecommons.org/licenses/by/4.0/), which permits unrestricted use, distribution and reproduction in any medium or format, provided the original work is properly cited. |
| Copyright |
Copyright © The Author(s), 2024. Published by Science Publishing Group |
Chlorine Demand, Private Borehole Water, Larson Corrosion Index, Germs Indicator of Faecal Contamination
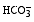 ) concentrations were determined via Mohr's method and titration with concentrated acid, respectively. Principal component analysis (PCA) (Origin Software) was used to highlight correlations between the various physicochemical parameters and understand the processes responsible for mineralization.
) concentrations were determined via Mohr's method and titration with concentrated acid, respectively. Principal component analysis (PCA) (Origin Software) was used to highlight correlations between the various physicochemical parameters and understand the processes responsible for mineralization. Parameters | Method | Equipment |
|---|---|---|
Color | Photometry | HANNA H197727 |
pH | Electrometry | WTW pH 3110/ SET2 |
Turbidity | Nephelometry | HACH 2100Qis |
Conductivity | Conductometry | WTW Cond 330i/SET |
TA - TAC | Titrimetric Method | - |
HCO3- | Titrimetric Method | - |
Ca2+ | Titrimetric Method | - |
Mg2+ | Titrimetric Method | - |
OxKMnO4 | Hot Method | - |
Cl- | Mohr's Method | - |
SO42- | Nephelometry | Molecular Absorption Spectrophotometer GENESYS UV‒Vis |
NH4+ | Nessler Method | Molecular Absorption Spectrophotometer GENESYS UV‒Vis |
NO2- | Spectrophotometry | Molecular Absorption Spectrophotometer GENESYS UV‒Vis |
NO3- | Spectrophotometry | Molecular Absorption Spectrophotometer GENESYS UV‒Vis |
FeTotal | Spectrophotometry | Molecular Absorption Spectrophotometer GENESYS UV‒Vis |
PO43- | Spectrophotometry | Molecular Absorption Spectrophotometer GENESYS UV‒Vis |
Silica | Spectrophotometry | Molecular Absorption Spectrophotometer GENESYS UV‒Vis |
Parameters | Method | Culture medium | Incubation (Temperature/Time) |
|---|---|---|---|
Total coliforms | Membrane filtration (0.45 µm) | Lauryl Sulfate | 37°C/24 h |
Faecal coliforms | Lauryl Sulfate | 44°C/24 h | |
Faecal Enterococci | Fresh Blood Agar | 37°C/24 h | |
Clostridium perfigens | Tryptone Sulfite Neomycin | 37°C/24 h |
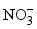 ,
, 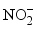 ,
, 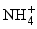 ), only nitrates have high concentrations, exceeding the related WHO standard. 17 samples against 13 have a concentration of
), only nitrates have high concentrations, exceeding the related WHO standard. 17 samples against 13 have a concentration of 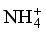 below the method limit of detection (MLD) of <0.001 mg/L in the dry and wet seasons, respectively. This trend is striking for
below the method limit of detection (MLD) of <0.001 mg/L in the dry and wet seasons, respectively. This trend is striking for 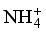 , where all values are below the MDL for both seasons.
, where all values are below the MDL for both seasons. Parameters | Dry season (n=32) | |||||||
|---|---|---|---|---|---|---|---|---|
Min | Max | Mean | StD | CV | WHO Standards (mg/L) | Not conformed groundwater sources | % out of standards range | |
Colour (mg/L PtU) | 0 | 112 | 15.47 | 22.68 | 146.63 | ≤ 15 | 8 (F1, F2, F4, F6, F8, F12, F21, F30) | 25 |
pH | 6.79 | 7.9 | 7.32 | 0.24 | 3.27 | 6.5 - 8.5 | 0 | 0 |
Turb (NTU) | 0.22 | 20.3 | 2.5 | 3.48 | 138.99 | < 5 | 5 (F2, F4, F6, F8, F30) | 15.52 |
Cond (µS/cm) | 219 | 2120 | 577.56 | 290.68 | 50.33 | 400 to 800 | 5 above 800 (F8, F9, F11, F25, F29) | 9.38 |
Mineralisation (mg/L) | 149.3 | 1499.84 | 411.81 | 200.56 | 48.7 | 1000 | 1 (F8) | 3.12 |
TA (°F) | 0 | 0 | 0 | 0 | NA | 0 | 0 | |
TAC (°F) | 5.4 | 33 | 18.85 | 4.41 | 23.41 | 5 to 30 | 2 (F11, F25) | 6.25 |
HCO3- (mg/L) | 65.88 | 402.6 | 230.01 | 53.84 | 23.41 | - | ||
Cl- (mg/L) | 17.75 | 191.7 | 55.34 | 30.51 | 55.13 | < 250 | 0 | 0 |
TH (°F) | 5.8 | 61 | 21.33 | 10.29 | 48.23 | ≤ 15 | 21 (only F3 to F7; F10, F16. F18, F28, F30, F31 are conform) | 65.63 |
Ca2+ (mg/l) | 16 | 1344 | 48.09 | 20.69 | 43.02 | - | ||
Mg2+ (mg/l) | 2.92 | 97.2 | 22.61 | 15.56 | 68.82 | - | ||
Oxydability (mgO2/L) | 0 | 2.15 | 0.25 | 0.36 | 146.43 | < 5 | 0 | 0 |
NH4+ (mg/L) | 0.04 | 0.06 | 0.05 | 0.01 | 12.23 | < 1.5 | 0 | 0 |
NO2- (mg/L) | < 0.001 | < 0.001 | < 0.001 | NA | NA | < 3 | 0 | 0 |
NO3- (mg/L) | 0.33 | 78.37 | 13.56 | 17.14 | 126.43 | < 50 | 3 (F12, F25, F29) | 9.38 |
FeT (mg/L) | 0.01 | 1.96 | 0.24 | 0.44 | 185.32 | < 0.3 | 3 (F8, F21, F30) | 9.38 |
SO42- (mg/L) | 1.91 | 157.33 | 22.6 | 20.45 | 90.49 | < 250 | 0 | 0 |
SO2 (mg/L) | 12.1 | 108.54 | 50.34 | 14.59 | 28.99 | |||
PO43- (mg/L) | 0.09 | 0.46 | 0.28 | 0.15 | 53 | |||
Parameters | Rainy Season (n=32) | |||||||
|---|---|---|---|---|---|---|---|---|
Min | Max | Mean | StD | CV | WHO Standards (µg/L) | Not conformed groundwater sources | % out of standards range | |
Colour (mg/L PtU) | 0 | 121 | 11.81 | 16.32 | 138.18 | ≤ 15 | 8 (F1, F2, F4, F6, F8, F12, F21, F30) | 25 |
pH | 6.8 | 7.9 | 7.4 | 0.2 | 2.7 | 6.5 - 8.5 | 0 | 0 |
Turb (NTU) | 0.14 | 21.2 | 1.96 | 2.72 | 138.63 | < 5 | 1 (F30) | 3.12 |
Cond (µS/cm) | 217 | 2180 | 598.56 | 291.51 | 48.7 | 400 to 800 | 5 above 800 (F8, F9, F11, F25, F29) | 9.38 |
Mineralisation (mg/L) | 149 | 1500 | 412 | 201 | 48.7 | 1000 | 1 (F8) | 3.12 |
TA (°F) | 0 | 0 | 0 | 0 | NA | |||
TAC (°F) | 6.4 | 31.8 | 17.23 | 4.36 | 25.31 | 5 to 30 | 1 (F26) | 3.12 |
HCO3- (mg/L) | 78.08 | 387.96 | 210.15 | 53.18 | 25.31 | - | ||
Cl- (mg/L) | 10.65 | 95.85 | 35 | 18.26 | 52.18 | < 250 | 0 | 0 |
TH (°F) | 4 | 68 | 25.21 | 10.25 | 40.65 | ≤ 15 | 25 (only F3 to F5; F7, F10, F28, F31 are conform) | 78.12 |
Ca2+ (mg/l) | 12 | 159.2 | 58.3 | 25.81 | 44.27 | |||
Mg2+ (mg/l) | 2.43 | 68.53 | 25.85 | 12.56 | 48.58 | |||
Oxydability (mgO2/L) | 0 | 2.42 | 0.56 | 0.4 | 71.04 | < 5 | 0 | 0 |
NH4+ (mg/L) | 0.03 | 0.07 | 0.04 | 0.01 | 18.83 | < 1.5 | 0 | 0 |
NO2- (mg/L) | < 0.001 | < 0.001 | < 0.001 | NA | NA | < 3 | 0 | 0 |
NO3- (mg/L) | 0.53 | 101.37 | 25.98 | 21.78 | 83.86 | < 50 | 6 (F9, F11, F12, F15, F25, F30) | 18.8 |
FeT (mg/L) | 0.01 | 2.12 | 0.28 | 0.48 | 172.27 | < 0.3 | 3 (F8, F21, F30) | 9.4 |
SO42- (mg/L) | 1.1 | 153.6 | 33.19 | 30.64 | 92.33 | < 250 | 0 | 0 |
SO2 (mg/L) | 7.9 | 83.03 | 40.34 | 17.57 | 43.55 | |||
PO43- (mg/L) | < 0.001 | < 0.001 | < 0.001 | NA | NA | 0 | 0 | |
Variables | Turb | Cond | Ca2+ | Mg2+ | FeT | NH4+ | HCO3- | Cl- | SO42- | NO3- | SiO2 |
|---|---|---|---|---|---|---|---|---|---|---|---|
Turb | 1 | (a) | |||||||||
Cond | 0,477 | 1 | |||||||||
Ca2+ | 0,330 | 0,763 | 1 | ||||||||
Mg2+ | 0,037 | 0,698 | 0,509 | 1 | |||||||
FeT | 0,759 | 0,576 | 0,470 | 0,151 | 1 | ||||||
NH4+ | 0,538 | -0,013 | -0,067 | -0,207 | 0,326 | 1 | |||||
HCO3- | -0,128 | 0,415 | 0,392 | 0,519 | -0,047 | -0,203 | 1 | ||||
Cl- | -0,028 | 0,703 | 0,485 | 0,615 | 0,092 | -0,246 | 0,501 | 1 | |||
SO42- | 0,569 | 0,754 | 0,589 | 0,216 | 0,721 | 0,086 | -0,049 | 0,340 | 1 | ||
NO3- | -0,277 | 0,313 | 0,215 | 0,646 | -0,182 | -0,096 | 0,203 | 0,407 | 0,032 | 1 | |
SiO2 | 0,151 | 0,202 | 0,124 | 0,291 | 0,253 | -0,129 | 0,124 | -0,063 | 0,171 | 0,130 | 1 |
Variables | Turb | Cond | Ca2+ | Mg2+ | FeT | NH4+ | HCO3- | Cl- | SO42- | NO3- | SiO2 |
|---|---|---|---|---|---|---|---|---|---|---|---|
Turb | 1 | ||||||||||
Cond | -0,037 | 1 | |||||||||
Ca2+ | 0,032 | 0,892 | 1 | (b) | |||||||
Mg2+ | -0,160 | 0,671 | 0,565 | 1 | |||||||
FeT | 0,486 | 0,546 | 0,441 | 0,393 | 1 | ||||||
NH4+ | 0,461 | 0,160 | 0,124 | -0,033 | 0,314 | 1 | |||||
HCO3- | -0,028 | 0,082 | 0,107 | 0,082 | 0,047 | 0,028 | 1 | ||||
Cl- | 0,003 | 0,879 | 0,884 | 0,555 | 0,461 | 0,059 | 0,085 | ||||
SO42- | -0,088 | 0,851 | 0,775 | 0,662 | 0,485 | 0,061 | 0,017 | 0,761 | 1 | ||
NO3- | -0,311 | 0,610 | 0,664 | 0,389 | -0,039 | -0,009 | -0,076 | 0,756 | 0,569 | 1 | |
SiO2 | -0,226 | 0,222 | 0,126 | 0,298 | 0,171 | -0,055 | -0,256 | 0,092 | 0,392 | 0,190 | 1 |
Total coliform (UFC/100 mL) | Thermotolerant coliform (UFC/100 mL) | Enterococcus faecalis (UFC/100 mL) | Clostridium perfigens (UFC/100 mL) | |||||
|---|---|---|---|---|---|---|---|---|
Dry season | Rainy season | Dry season | Rainy season | Dry season | Rainy season | Dry season | Rainy season | |
Min (UFC/100 mL) | 0,00 | 0,00 | 0,00 | 0,00 | 0,00 | 0,00 | 0,00 | 0,00 |
Max (UFC/100 mL) | 2240,0 | 5500,00 | 0,00 | 0,00 | 1200,00 | 1500,00 | 40,00 | 0,00 |
Mean (UFC/100 mL) | 193,44 | 737,19 | 0,00 | 0,00 | 226,25 | 410,31 | 1,25 | 0,00 |
STD | 469,04 | 1289,52 | 0,00 | 0,00 | 367,40 | 525,29 | 7,07 | 0,00 |
CV% | 2,42 | 1,75 | NA | NA | 1,62 | 1,28 | 5,66 | NA |
% of contaminated sample | 62,5 | 90,63 | 0 | 0 | 62,5 | 81,25 | 3,13 | 0 |
Chlorine demand (mg/L) | ||
|---|---|---|
Dry season | Rainy season | |
Max | 6,00 | 2,40 |
Min | 0,25 | 0,25 |
Mean | 0,82 | 0,93 |
Std | 1,08 | 0,45 |
CV (%) | 1,32 | 0,48 |
GDWQ | WHO Guidelines for Drinking Water Quality |
DBP | Disinfection Byproducts |
LR | Larson Corrosion Index |
WHO | World Health Organization |
TdE | Togolese Water Company |
| [1] | Hounsounou, E. O., Agassounon Djikpo Tchibozo, M., Kelome, N. C., Vissin, E. W., Mensah, G. A. et Agbossou, E. (2017) Pollution des eaux à usages domestiques dans les milieux urbains défavorisés des pays en développement : Synthèse bibliographique. International Journal of Biological and Chemical Sciences, 10, 2392. |
| [2] | Dougna, A. A., Gnazou, M. D.-T., Kodom, T., Djaneye-Boundjou, G. et Bawa, M. L. (2014) Physico-chimie et qualité des eaux des forages d’hydraulique villageoise dans la région centrale au Togo. International Journal of Biological and Chemical Sciences, 9, 2249. |
| [3] | Grondin, P. M. (2005) Chloration en milieu rural dans les pays en voie de développement [Internet]. Programme Solidarité Eau. |
| [4] | Rice, E. W., Bridgewater, L., APHA, AWWA et WEF. (2012) Standard Methods for the Examination of Water and Wastewater [Internet]. American Public Health Association. |
| [5] | WHO. (2022) Guidelines for drinking-water quality. Fourth edition incorporating the first and second addenda. World Health Organization, Geneva. |
| [6] | Nielsen, A. M., Garcia, L. A. T., Silva, K. J. S., Sabogal-Paz, L. P., Hincapié, M. M., Montoya, L. J. et al. (2022) Chlorination for low-cost household water disinfection – A critical review and status in three Latin American countries. International Journal of Hygiene and Environmental Health, 244, 114004. |
| [7] | Jandova, J., Schiro, G., Duca, F. A., Laubitz, D. et Wondrak, G. T. (2024) Exposure to chlorinated drinking water alters the murine feacal microbiota. Science of The Total Environment, 914, 169933. |
| [8] | Lacina, C., Dramane, D., Adama, C. et Germain, G. (2004) Utilisation des ressources en eaux, assainissement et risques sanitaires dans les quartiers précaires de la commune de Port-Bouët (Abidjan; Côte d’Ivoire). VertigO, |
| [9] | Primature. (2021) Phase 2 du PASSCO, de l’eau potable dans la Kara et les Savanes à travers 600 forages [Internet]. |
| [10] | ATOP. (2023) Société / récurrence du problème d’eau à Kara : les mesures alternatives [Internet]. |
| [11] | Adewi, E. et Dubreuil, V. (2012) Variabilité Climatique et paludisme à Kara, une ville du Nord-Togo. 25ème Colloque de l’Association Internationale de Climatologie, Grenoble. |
| [12] | Bawa, L. M., Tchakala, I. et Djaneye-Boundjou, G. (2008) Détermination de la demande en chlore des eaux de puits et de forages d’un quartier périurbain de la ville de Lomé : incidence sur la désinfection. Journal des Sciences et Technologie, 7, 19‑24. |
| [13] | Tampo, L., Ayah, M., Kodom, T., Tchakakla, I., Boguido, P., Bawa, L. et al. (2014) Impact de la demande en chlore et de la chloration sur la désinfection des eaux de puits des quartiers de Lomé : cas des quartiers de Démakpoé et d’Agbalépédogan (Togo). Journal of Applied Biosciences, 75, 6272. |
| [14] | Segbeaya, K. N. (2012) Evaluation de l’impact des déchets ménagers de la ville de Kara (Togo) sur la qualité de la rivière Kara. Université de Lomé et Limoges. |
| [15] | INSEED. (2023) Présentation des principaux résultats définitifs du RGPH-5 [Internet]. |
| [16] | Fandjinou, K. (2019) Analysis of climate variability and its relations to vegetation dynamics in Togo, Western Africa from 1984 to 2017. Applied Ecology and Environmental Research, 17. |
| [17] | INSEED. (2023) Annuaire statistique de la région de la Kara de 2020 et 2021 [Internet]. |
| [18] | Mairie de Kara. (2018) Plan de Développement Communal de Kara 2019 - 2023. |
| [19] | Tairou, M. S. et Affaton, P. (2013) Structural Organization and Tectono-Metamorphic Evolution of the Pan-African Suture Zone: Case of the Kabye and Kpaza Massifs in the Dahomeyide Orogen in Northern Togo (West Africa). International Journal of Geosciences, 04, 166‑82. |
| [20] | Rodier, J., Legube, B., Merlet, N. et Brunet, R. (2009) L’analyse de l’eau - 9e éd.: Eaux naturelles, eaux résiduaires, eau de mer. Dunod. |
| [21] | Chippaux, J.-P., Houssier, S., Gross, P., Bouvier, C. et Brissaud, F. (2002) Etude de la pollution de l’eau souterraine de la ville de Niamey, Niger. Bulletin de la Société de Pathologie Exotique, 95, 119‑23. |
| [22] | Owolabi, R. U. et Azeez, L. A. (2010) Transit Monitoring of Residual Chlorine in Awe-Oyo Area of Nigeria Water Township Supply. International Journal of Chemical Engineering and Applications, 143‑6. |
| [23] | Psakis, G., Spiteri, D., Mallia, J., Polidano, M., Rahbay, I. et Valdramidis, V. P. (2023) Evaluation of Alternative-to-Gas Chlorination Disinfection Technologies in the Treatment of Maltese Potable Water. Water, 15, 1450. |
| [24] | Shikongo-Nambabi, M. N.-N. N., Barbara, K. et Stephanus N, V. (2010) Evaluation of oxidising disinfectants to control Vibrio biofilms in treated seawater used for fish processing. Water SA, 36, 215‑20. |
| [25] | Abascal, E., Gómez-Coma, L., Ortiz, I. et Ortiz, A. (2022) Global diagnosis of nitrate pollution in groundwater and review of removal technologies. Science of The Total Environment, 810, 152233. |
| [26] | Lapworth, D. J., Nkhuwa, D. C. W., Okotto-Okotto, J., Pedley, S., Stuart, M. E., Tijani, M. N. et al. (2017) Urban groundwater quality in sub-Saharan Africa: current status and implications for water security and public health. Hydrogeology Journal, 25, 1093‑116. |
| [27] | Rajkumar, Y. et Xu, Y. (2011) Protection of Borehole Water Quality in Sub-Saharan Africa using Minimum Safe Distances and Zonal Protection. Water Resources Management, 25, 3413‑25. |
| [28] | Law n°2010 - 004 on the Water Code [Internet]. |
| [29] | Legube, B. (2021) Production d’eau potable: procédés de traitement, paramètres de qualité et impacts du changement climatique. 2e éd. Dunod, Malakoff. |
| [30] | Bradberry, S. (2016) Methaemoglobinaemia. Medicine, 44, 91‑2. |
| [31] | Hou, P., Jiang, Y., Yan, L., Petropoulos, E., Wang, J., Xue, L. et al. (2021) Effect of fertilization on nitrogen losses through surface runoffs in Chinese farmlands: A meta-analysis. Science of The Total Environment, 793, 148554. |
| [32] | Wu, S., Bashir, M. A., Raza, Q.-U.-A., Rehim, A., Geng, Y. et Cao, L. (2023) Application of riparian buffer zone in agricultural non-point source pollution control—A review. Frontiers in Sustainable Food Systems, 7, 985870. |
| [33] | Rahoui, M., Soudi, B. et Ahmad, F. I. (2000) Situation actuelle de la pollution nitrique des eaux souterraines dans le périmètre irrigué des Doukkala [Internet]. Transfert de Technologie en Agriculture. |
| [34] | Zhang, H., Liu, D., Zhao, L., Wang, J., Xie, S., Liu, S. et al. (2022) Review on corrosion and corrosion scale formation upon unlined cast iron pipes in drinking water distribution systems. Journal of Environmental Sciences, 117, 173‑89. |
| [35] | Ahmed, S., Sultan, M. W., Alam, M., Hussain, A., Qureshi, F. et Khurshid, S. (2021) Evaluation of corrosive behavior and scaling potential of shallow water aquifer using corrosion indices and geospatial approaches in regions of the Yamuna river basin. Journal of King Saud University - Science, 33, 101237. |
| [36] | Ozair, G. (2012) An Overview of Calcium Carbonate Saturation Indices as a Criterion to Protect Desalinated Water Transmission Lines From Deterioration. Nature Environment and Pollution Technology, 11, 203‑12. |
| [37] | Marconnet, C., Dagbert, C., Roy, M. et Féron, D. (2005) Comportement d’aciers inoxydables en eaux naturelles. Matériaux & Techniques, 93, s. 83-s. 90. |
| [38] | Akpataku, K. V., Gnazou, M. D. T., Djanéyé-Boundjou, G., Bawa, L. M. et Faye, S. (2020) Role of Natural and Anthropogenic Influence on the Salinization of Groundwater from Basement Aquifers in the Middle Part of Mono River Basin, Togo. Journal of Environmental Protection, 11, 1030‑51. |
| [39] | Gnazou, M., Assogba, K., Sabi, B. et Bawa, L. (2015) Qualité physico-chimique et bactériologique des eaux utilisées dans les écoles de la préfecture de Zio (Togo). International Journal of Biological and Chemical Sciences, 9, 504. |
| [40] | Akpataku, K. V., Gnazou, M. D. T., Nomesi, T. Y. A., Nambo, P., Bawa, L. M. et Djaneye-Boundjou, G. (2020) Physicochemical and Microbiological Quality of Shallow Groundwater in Lomé, Togo. Journal of Geoscience and Environment Protection, 08, 162‑79. |
| [41] | Soncy, K., Djeri, B., Anani, K., Eklou-Lawson, M., Adjrah, Y., Karou, D. et al. (2015) Évaluation de la qualité bactériologique des eaux de puits et de forage à Lomé, Togo. Journal of Applied Biosciences, 91, 8464. |
| [42] | Kamanula, J. F., Zambasa, O. J. et Masamba, W. R. L. (2014) Quality of drinking water and cholera prevalence in Ndirande Township, City of Blantyre, Malawi. Physics and Chemistry of the Earth, Parts A/B/C, 72‑75, 61‑7. |
| [43] | MacKeown, H., Von Gunten, U. et Criquet, J. (2022) Iodide sources in the aquatic environment and its fate during oxidative water treatment – A critical review. Water Research, 217, 118417. |
| [44] | WHO et FAO. (2009) Principles and methods for the risk assessment of chemicals in food. World Health Organization, World Health Organization, Geneva. 18. |
| [45] | Lundgren, R. E. et McMakin, A. H. (2013) Risk communication: a handbook for communicating environmental, safety, and health risks. 5e ed. Wiley IEEE press, Hoboken Piscataway. |
APA Style
Dougna, A. A., Kolani, J. M., Akpataku, K. V., Tchakala, I., Tampo, L., et al. (2024). Quality and Chlorine Demand of Private Raw Borehole Water in the City of Kara, Togo. American Journal of Applied Chemistry, 12(6), 158-172. https://doi.org/10.11648/j.ajac.20241206.14
ACS Style
Dougna, A. A.; Kolani, J. M.; Akpataku, K. V.; Tchakala, I.; Tampo, L., et al. Quality and Chlorine Demand of Private Raw Borehole Water in the City of Kara, Togo. Am. J. Appl. Chem. 2024, 12(6), 158-172. doi: 10.11648/j.ajac.20241206.14
AMA Style
Dougna AA, Kolani JM, Akpataku KV, Tchakala I, Tampo L, et al. Quality and Chlorine Demand of Private Raw Borehole Water in the City of Kara, Togo. Am J Appl Chem. 2024;12(6):158-172. doi: 10.11648/j.ajac.20241206.14
@article{10.11648/j.ajac.20241206.14,
author = {Akpénè Amenuvevega Dougna and Jeff Milibe Kolani and Kossitse Venyo Akpataku and Ibrahim Tchakala and Lallébila Tampo and Alfa-Sika Mande Seyf-Laye and Moctar Limam Bawa},
title = {Quality and Chlorine Demand of Private Raw Borehole Water in the City of Kara, Togo
},
journal = {American Journal of Applied Chemistry},
volume = {12},
number = {6},
pages = {158-172},
doi = {10.11648/j.ajac.20241206.14},
url = {https://doi.org/10.11648/j.ajac.20241206.14},
eprint = {https://article.sciencepublishinggroup.com/pdf/10.11648.j.ajac.20241206.14},
abstract = {The reliance on borehole water necessitates a thorough understanding of its quality. Due to water shortage, many households in the city of Kara use water from boreholes directly supplied untreated in the dry and wet seasons. Therefore, this work aimed to investigate the chlorine demand of raw water from private boreholes and perform a spatiotemporal evaluation of the water quality in the city of Kara, Togo. Two sampling campaigns spread over dry (DS) and rainy (RS) seasons in April and August 2023, were carried out to capture seasonal and spatial differences across various boreholes (n=32) in the study area. The physicochemical parameters and germ indicators of fecal contamination were assessed via the standardized method (AFNOR) and WHO Guidelines for Drinking Water Quality (GDWQ). The correlation matrix analysis revealed a complex effect from the dissolution of local rocks (amphibolites, pyroxenites, etc.) and anthropogenic activities. Water was noncompliant concerning faecal contamination in 84.3% of the cases in the dry season (DS) and 96.37% of the cases in the rainy season (RS). The color, TH, TAC, turbidity, FeT, nitrate ion, and electrical conductivity values for some boreholes were outside the GDWQ values. Boreholes F8, F9, and F25 have very high Larson corrosion index (LR>1.2). The chlorine demand varies according to the season, ranging from 0.25 to 6.0 mg/L in DS and from 0.25 to 2.45 mg/L in RS. Regular monitoring is needed to ensure safe drinking water from boreholes in Kara.
},
year = {2024}
}
TY - JOUR T1 - Quality and Chlorine Demand of Private Raw Borehole Water in the City of Kara, Togo AU - Akpénè Amenuvevega Dougna AU - Jeff Milibe Kolani AU - Kossitse Venyo Akpataku AU - Ibrahim Tchakala AU - Lallébila Tampo AU - Alfa-Sika Mande Seyf-Laye AU - Moctar Limam Bawa Y1 - 2024/12/27 PY - 2024 N1 - https://doi.org/10.11648/j.ajac.20241206.14 DO - 10.11648/j.ajac.20241206.14 T2 - American Journal of Applied Chemistry JF - American Journal of Applied Chemistry JO - American Journal of Applied Chemistry SP - 158 EP - 172 PB - Science Publishing Group SN - 2330-8745 UR - https://doi.org/10.11648/j.ajac.20241206.14 AB - The reliance on borehole water necessitates a thorough understanding of its quality. Due to water shortage, many households in the city of Kara use water from boreholes directly supplied untreated in the dry and wet seasons. Therefore, this work aimed to investigate the chlorine demand of raw water from private boreholes and perform a spatiotemporal evaluation of the water quality in the city of Kara, Togo. Two sampling campaigns spread over dry (DS) and rainy (RS) seasons in April and August 2023, were carried out to capture seasonal and spatial differences across various boreholes (n=32) in the study area. The physicochemical parameters and germ indicators of fecal contamination were assessed via the standardized method (AFNOR) and WHO Guidelines for Drinking Water Quality (GDWQ). The correlation matrix analysis revealed a complex effect from the dissolution of local rocks (amphibolites, pyroxenites, etc.) and anthropogenic activities. Water was noncompliant concerning faecal contamination in 84.3% of the cases in the dry season (DS) and 96.37% of the cases in the rainy season (RS). The color, TH, TAC, turbidity, FeT, nitrate ion, and electrical conductivity values for some boreholes were outside the GDWQ values. Boreholes F8, F9, and F25 have very high Larson corrosion index (LR>1.2). The chlorine demand varies according to the season, ranging from 0.25 to 6.0 mg/L in DS and from 0.25 to 2.45 mg/L in RS. Regular monitoring is needed to ensure safe drinking water from boreholes in Kara. VL - 12 IS - 6 ER -